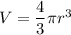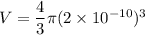Physics, 18.12.2019 04:31 silviamgarcia
The size (radius) of an oxygen molecule is about 2.0 ×10−10 m. make a rough estimate of the pressure at which the finite volume of the molecules should cause noticeable deviations from ideal-gas behavior at ordinary temperatures (t= 300k ). assume that deviatons would be noticeable when volume of the gas per molecule equals the volume of the molecule itself.
express your answer using one significant figure.
p = ? pa

Answers: 3
Another question on Physics

Physics, 22.06.2019 09:30
On a day when the barometer reads 75.23 cm, a reaction vessel holds 250 ml of ideal gas at 20 celsius. an oil manometer ( ρ= 810 kg/m^3) reads the pressure in the vessel to be 41 cm of oil and below atmospheric pressure. what volume will the gas occupy under s.t.p.?
Answers: 2

Physics, 22.06.2019 10:30
In automobile steering wheel is shown what is the ideal mechanical advantage blank? if the ama is eight what is the efficiency of the steering wheel?
Answers: 1

Physics, 22.06.2019 16:30
In a classical model of the hydrogen atom, the electron moves around the proton in a circular orbit of radius 0.053 nm. what is the electron's orbital frequency? what is the effective current of the electron?
Answers: 3

Physics, 22.06.2019 16:30
Write a polynomial function with the given zeros x= -1,0,2show your work
Answers: 3
You know the right answer?
The size (radius) of an oxygen molecule is about 2.0 ×10−10 m. make a rough estimate of the pressure...
Questions


Mathematics, 09.02.2020 23:13

Mathematics, 09.02.2020 23:13

Mathematics, 09.02.2020 23:13

History, 09.02.2020 23:13

Biology, 09.02.2020 23:13

Physics, 09.02.2020 23:13



Business, 09.02.2020 23:14

History, 09.02.2020 23:14

Chemistry, 09.02.2020 23:14

English, 09.02.2020 23:14



Computers and Technology, 09.02.2020 23:14


Computers and Technology, 09.02.2020 23:14


Mathematics, 09.02.2020 23:15