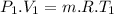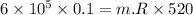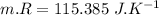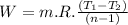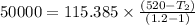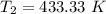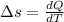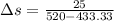Nitrogen (n2) undergoes an internally reversible process from 6 bar, 247°c during which pν1.2 = constant. the initial volume is 0.1 m3 and the work for the process is 50 kj. assuming ideal gas behavior, and neglecting kinetic and potential energy effects, determine heat transfer, in kj, and the entropy change, in kj/k.

Answers: 2
Another question on Physics

Physics, 22.06.2019 05:30
Will give brainliest! which statement best describes the difference between strong nuclear forces and weak nuclear forces? weak nuclear forces are involved when certain types of atoms break down. strong nuclear forces are responsible for holding atoms' nucleus together. weak nuclear forces hold bonds between atoms together. strong nuclear forces hold together the nucleus of an atom. strong nuclear bonds prevent atoms from falling apart. weak nuclear bonds prevent compounds from falling apart. strong nuclear forces are involved in breaking electrons from their shells. weak nuclear forces hold protons in the nucleus.
Answers: 3

Physics, 22.06.2019 09:30
Need asap ‼️ 20 pts which gravitational force field diagram is drawn correctly? (answers in pictures below)
Answers: 1

Physics, 22.06.2019 10:00
Students design a model roller-coaster track. they place a rubber ball at the highest point on the track and let it go. the ball rolls along the track pulled only by the force of gravity. eventually, it comes to a stop. which change to the design will result in the ball moving the greatest distance?
Answers: 1

Physics, 22.06.2019 11:30
If the chemical properties of a substance remain unchanged and appearance or shape of a substance changes it is called a ?
Answers: 1
You know the right answer?
Nitrogen (n2) undergoes an internally reversible process from 6 bar, 247°c during which pν1.2 = cons...
Questions


Biology, 01.08.2019 05:30




Mathematics, 01.08.2019 05:30




Mathematics, 01.08.2019 05:30

Mathematics, 01.08.2019 05:40

History, 01.08.2019 05:40

Mathematics, 01.08.2019 05:40

History, 01.08.2019 05:40

History, 01.08.2019 05:40

History, 01.08.2019 05:40

History, 01.08.2019 05:40

Social Studies, 01.08.2019 05:40


History, 01.08.2019 05:40

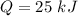
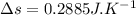
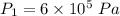 initial temperature,
initial temperature, 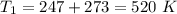 polytropic index,
polytropic index, 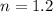 initial volume,
initial volume, 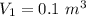 work done in the process,
work done in the process, 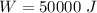
![Q=W[\frac{\gamma -n}{\gamma-1} ]](/tpl/images/0402/4036/a3071.png)
![Q=50\times[\frac{1.4-1.2}{1.4-1} ]](/tpl/images/0402/4036/3ed0d.png)