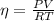The volume of an ideal gas is adiabatically reduced from 151 l to 80.6 l. the initial pressure and temperature are 1.50 atm and 260 k. the final pressure is 3.61 atm. (a) is the gas monatomic, diatomic, or polyatomic? (b) what is the final temperature? (c) how many moles are in the gas?

Answers: 3
Another question on Physics

Physics, 22.06.2019 16:50
Which best describes the first law of thermodynamics as compared to the second law of thermodynamics? a. the first law describes how thermal energy is conserved but not the direction it moves. b. the first law describes the direction thermal energy moves but not how it is conserved. c. the first law describes how thermal energy can be created but not how it can be destroyed. d. the first law describes how thermal energy can be destroyed but not how it can be created.
Answers: 1

Physics, 22.06.2019 19:30
In this thread, i would like you to comment on the nature of light and how operation of telescopes. light has a duality of a particle and a wave; which one affects your life? also how does light interact with optics inside telescopic systems? answer these questions in two paragraphs. then respond the another students response.
Answers: 1

Physics, 22.06.2019 19:40
12. a body is a particular amount of matter. it can be a solid, liquid or gas. it can be described as existing in a. size and shape. b. motion and force c. time and space. d. location and movement.
Answers: 2

Physics, 22.06.2019 22:00
Which statement best explains why the overall charge on an atom is zero?
Answers: 1
You know the right answer?
The volume of an ideal gas is adiabatically reduced from 151 l to 80.6 l. the initial pressure and t...
Questions





Computers and Technology, 21.04.2020 21:49





Mathematics, 21.04.2020 21:49

Computers and Technology, 21.04.2020 21:49

Spanish, 21.04.2020 21:49

Mathematics, 21.04.2020 21:49



Mathematics, 21.04.2020 21:49

Advanced Placement (AP), 21.04.2020 21:49


Social Studies, 21.04.2020 21:49

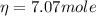
 value
value

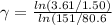
](/tpl/images/0110/3620/2a67e.png)
![T_f = 260*[\frac{151}{80.6}]^ {(1.38-1)}](/tpl/images/0110/3620/31191.png)