
Answers: 2
Another question on Physics

Physics, 21.06.2019 18:20
The extremely high temperatures needed to trigger nuclear fusion are proposed to be generated by laser irradiating a spherical pellet of deuterium and tritium fuel of diameter 1.8 mm. (a) determine the maximum fuel temperature that can be achieved by irradiating the pellet with 200 lasers, each producing a power of 550 w. the pellet has an absorptivity 0.3 and emissivity 0.8.
Answers: 2


Physics, 22.06.2019 16:50
Two loudspeakers, 5.5 m apart and facing each other, play identical sounds of the same frequency. you stand halfway between them, where there is a maximum of sound intensity. moving from this point toward one of the speakers, you encounter a minimum of sound intensity when you have moved 0.33 m . assume the speed of sound is 340 m/s.part a) what is the frequency of the sound? part b) if the frequency is then increased while you remain 0.21 m from the center, what is the first frequency for which that location will be a maximum of sound intensity? express your answer to two significant figures and include the appropriate units.
Answers: 2

Physics, 23.06.2019 02:30
Substances that assume the shape of their container, but do not have a definite size are called
Answers: 2
You know the right answer?
Which subatomic particle has a negative charge...
Questions

Mathematics, 12.11.2020 22:50


Business, 12.11.2020 22:50

Mathematics, 12.11.2020 22:50

Computers and Technology, 12.11.2020 22:50




Biology, 12.11.2020 22:50

Mathematics, 12.11.2020 22:50

SAT, 12.11.2020 22:50

Mathematics, 12.11.2020 22:50


Mathematics, 12.11.2020 22:50

Mathematics, 12.11.2020 22:50

Mathematics, 12.11.2020 22:50


History, 12.11.2020 22:50


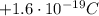 ), and a mass of approximately
), and a mass of approximately 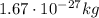
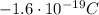 ), and their mass is about 1800 time smaller than the mass of the proton (
), and their mass is about 1800 time smaller than the mass of the proton (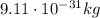 )
)

