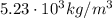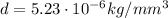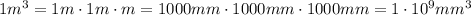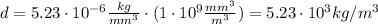In a laboratory, you determine that the density of a certain solid is 5.23×10−6kg/mm3. convert this density into kilograms per cubic meter. notice that the units you are trying to eliminate are now in the denominator. the same principle from the previous parts applies: pick the conversion factor so that the units cancel. the only change is that now the units you wish to cancel must appear in the numerator of the conversion factor.

Answers: 3
Another question on Physics

Physics, 21.06.2019 17:20
Your 249 ml cup of coffee is too hot to drink when served at 82 degree celsius. what is the mass of an ice cube taken from a -21 degree celsius freezer, that will cool your coffee to a pleasant 54 degree celsius. specific heat of the water is 4190 j/kg c, specific heat of ice is 2090 j/kg c, heat of fusion of ice is 3.33 x 10^5 j/kg
Answers: 3

Physics, 22.06.2019 00:30
There are weak or strong attractive forces between atoms of liquids with a high viscosity
Answers: 3

Physics, 22.06.2019 06:30
The energy of a photon was found to be 3.38 × 10–19 j. planck’s constant is 6.63 × 10–34 j • s. which color of light corresponds to this photon?
Answers: 2

You know the right answer?
In a laboratory, you determine that the density of a certain solid is 5.23×10−6kg/mm3. convert this...
Questions




Mathematics, 02.12.2020 22:40


Mathematics, 02.12.2020 22:40

Mathematics, 02.12.2020 22:40



Mathematics, 02.12.2020 22:40



Physics, 02.12.2020 22:40


Mathematics, 02.12.2020 22:40


Mathematics, 02.12.2020 22:40

Mathematics, 02.12.2020 22:40


Biology, 02.12.2020 22:40