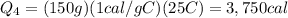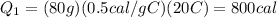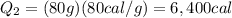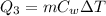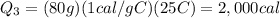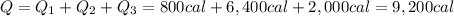The initial temperature of 150 g of ice is °c. the spe- cific heat capacity of ice is 0.5 cal/g·c° and water’s is 1 cal/g·c°. the latent heat of fusion of water is 80 cal/g. a. how much heat is required to raise the ice to 0°c and completely melt the ice? b. how much additional heat is required to heat the water (obtained by melting the ice) to 25°c? c. what is the total heat that must be added to convert the 80 g of ice at °c to water at °c? d. can we find this total heat simply by computing how much heat is required to melt the ice and adding the amount of heat required to raise the temperature of 80 g of water by 45°c? explain.

Answers: 3
Another question on Physics

Physics, 22.06.2019 03:30
Calculate the mass of an object that has a momentum of 100kg x m/sec and velocity of 4 m/sec
Answers: 1

Physics, 22.06.2019 08:30
If an astronaut takes an object to the moon the following is true (choose only one). a the mass will change but the weight will be the same. b both the mass and the weight of the object will be the same. c the weight will change but the mass will be the same. d both the mass and the weight of the object will change.
Answers: 1

Physics, 22.06.2019 11:30
While you are driving in the lane next to the curb on a multi-lane road the car on your left suddenly moves toward you lane. they are about toy crash into your front fender. you
Answers: 2

You know the right answer?
The initial temperature of 150 g of ice is °c. the spe- cific heat capacity of ice is 0.5 cal/g·c° a...
Questions


English, 08.06.2021 18:50





Mathematics, 08.06.2021 18:50

Mathematics, 08.06.2021 18:50


Mathematics, 08.06.2021 18:50

Biology, 08.06.2021 18:50


History, 08.06.2021 18:50

Mathematics, 08.06.2021 18:50


Mathematics, 08.06.2021 18:50

Mathematics, 08.06.2021 18:50


Biology, 08.06.2021 18:50

Mathematics, 08.06.2021 18:50

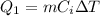
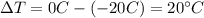 is the change in temperature of the ice
is the change in temperature of the ice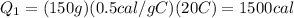
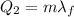
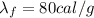 is the latent heat of fusion
is the latent heat of fusion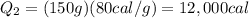
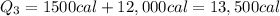
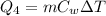
 is the change in temperature
is the change in temperature