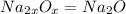Answers: 3
Another question on Physics

Physics, 22.06.2019 00:10
The energy released by a chemical reaction can be measured using a calorimeter. when barium hydroxide octahydrate crystals are reacted with dry ammonium chloride inside of a coffee cup calorimeter, the temperature of the 18.00 g of water in the calorimeter decreases from 30.0°c to 8.0°c. the equation for calculating energy absorbed or released by a reaction is: where q is the energy released or absorbed, m is the mass of water in the calorimeter, cp is the specific heat of water, and δt is the observed temperature change. if the specific heat of liquid water is 4.19 j/g·°c, how much energy was absorbed by the reaction?
Answers: 3



Physics, 22.06.2019 16:30
An astronaut in space cannot use a scale or balance to weigh objects because there is no gravity. but she does have devices to measure distance and time accurately. she knows her own mass is 77.4 kg , but she is unsure of the mass of a large gas canister in the airless rocket. when this canister is approaching her at 3.50 m/s , she pushes against it, which slows it down to 1.30 m/s (but does not reverse it) and gives her a speed of 2.60 m/s . what is the mass of the canister?
Answers: 1
You know the right answer?
The ratio of na to o is 2: 1. what is the chemical formula for this ionic compound? nao nao2 na2o n...
Questions




Chemistry, 21.09.2021 19:10




Chemistry, 21.09.2021 19:10

Mathematics, 21.09.2021 19:10



Computers and Technology, 21.09.2021 19:10

Business, 21.09.2021 19:10



Chemistry, 21.09.2021 19:10


Chemistry, 21.09.2021 19:10

Mathematics, 21.09.2021 19:10

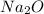 .
.