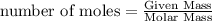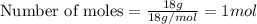Physics, 29.06.2019 22:00 jaidenkenna2001
Specific heat of water = 4.186 j g°c specific heat of ice = 2.00 j g°c molar heat of fusion = 6030 j mol molar heat of vaporization = 40790 j mol you take an ice cube (mass = 18g) from the freezer (t = -10°c) and place it on the table. later that day, you notice a puddle of water on the table that has reached ambient room temperature (20°c). how much heat must have been absorbed to make this happen?

Answers: 1
Another question on Physics

Physics, 21.06.2019 21:30
Janelle made a hypothesis bout the uneven temperatures inside her house during winter. she believes that 50% of the ducts are blocked and she plans to investigate. if she wants to prove her hypothesis using scientific processes what should she do next?
Answers: 2

Physics, 21.06.2019 21:50
Acar travels along a highway with a velocity of 24 m/s, west. the car exits the highway; and 4.0 s later, its instantaneous velocity is 16 m/s, 45° north of west. what is the magnitude of the average acceleration of the car during the four-second interval?
Answers: 2


Physics, 22.06.2019 17:40
Aspring is used to stop a 50-kg package which is moving down a 20º incline. the spring has a constant k = 30 kn/m and is held by cables so that it is initially compressed 50 mm. knowing that the velocity of the package is 2 m/s when it is 8 m from the spring and neglecting friction, determine the maximum additional deformation of the spring in bringing the package to rest.
Answers: 1
You know the right answer?
Specific heat of water = 4.186 j g°c specific heat of ice = 2.00 j g°c molar heat of fusion = 6030...
Questions



Mathematics, 22.11.2020 20:50


History, 22.11.2020 20:50

English, 22.11.2020 20:50

English, 22.11.2020 20:50


Business, 22.11.2020 20:50

Mathematics, 22.11.2020 20:50

History, 22.11.2020 20:50


Computers and Technology, 22.11.2020 20:50





Mathematics, 22.11.2020 20:50

Mathematics, 22.11.2020 20:50