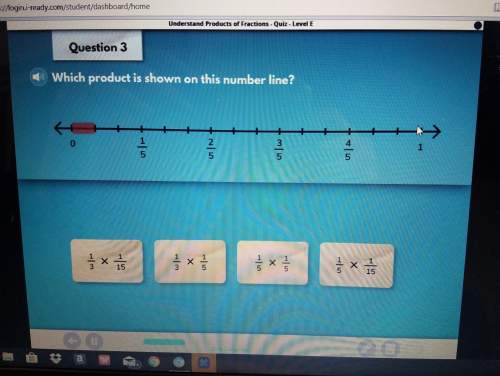
Mathematics, 04.02.2021 01:00 priceb17
A sample of H2SO4 contains 2.02 g of hydrogen, 32.07 g of sulfur, and 64.00 g of oxygen. How many grams of sulfur and grams of oxygen are present in a
second sample of H2SO4 containing 5.47 g of hydrogen?
Mass of sulfur = 8
Mass of oxygen
8
Submit Answer
Try Another Version
3 item attempts remaining

Answers: 3
Another question on Mathematics

Mathematics, 21.06.2019 17:40
The weight of full–grown tomatoes at a farm is modeled by a normal distribution with a standard deviation of 18.4 grams. the 95 percent confidence interval for the mean weight of the tomatoes is calculated using a sample of 100 tomatoes. what is the margin of error (half the width of the confidence interval)?
Answers: 2

Mathematics, 21.06.2019 19:00
What is the expression in factored form? 225x^2 - 144 a. 9(5x+4)^2 b. 9(5x-4)^2 c. 9(4x+5)(4x-5) d. 9(5x+4)(5x-4)
Answers: 2

Mathematics, 21.06.2019 22:00
Determine the domain and range of the given function. the domain is all real numbers all real numbers greater than or equal to –2{x: x = –2, –1, 0, 1, 2}{y: y = –2, –1, 0, 1, 2}. the range is all real numbers all real numbers greater than or equal to –2{x: x = –2, –1, 0, 1, 2}{y: y = –2, –1, 0, 1, 2}.
Answers: 1

You know the right answer?
A sample of H2SO4 contains 2.02 g of hydrogen, 32.07 g of sulfur, and 64.00 g of oxygen. How many gr...
Questions




History, 09.09.2020 22:01


Mathematics, 09.09.2020 22:01


Mathematics, 09.09.2020 22:01


English, 09.09.2020 22:01



English, 09.09.2020 22:01


Chemistry, 09.09.2020 22:01


Mathematics, 09.09.2020 22:01

Chemistry, 09.09.2020 22:01