
Mathematics, 24.12.2019 20:31 llamasking
50 pts and bra8inliest if u get it right and answer all
question 1 options:
indicate whether the reaction is an example of a synthesis, decomposition, combustion, single displacement or double displacement reaction. na3po4+3kohànaoh+k3po4
i think it might be double displacement but im not sure
question 2
name the reactants in the reaction from question 1. separate the reactants with a comma. do not use subscripts. for example, if the reactants are nh3 and h2o the answer would be entered as: nh3, h2o
i think its napo4 but im not sure
question 3
name the products in the reaction from question 1. separate the products with a comma. do not use subscripts. for example, if the products are nh3 and h2o the answer would be entered as: nh3, h2o
i think its naoh, k3po4 but im not sure
question 4
indicate whether the reaction is an example of a synthesis, decomposition, combustion, single displacement or double displacement reaction. c6h12+9o2à6co2+6h2o
question 5
name the reactants in the reaction from question 4. separate the reactants with a comma. do not use subscripts. for example, if the reactants are nh3 and h2o the answer would be entered as: nh3, h2o
i think its c6h12o6, 6o2 but im not sure
question 6
name the products in the reaction from question 4. separate the products with a comma. do not use subscripts. for example, if the products are nh3 and h2o the answer would be entered as: nh3, h2o
i think its 6co2,6h2o but im not sure
question 7
indicate whether the reaction is an example of a synthesis, decomposition, combustion, single displacement or double displacement reaction. pb+feso4àpbso4+fe
question 8 (
indicate whether the reaction is an example of a synthesis, decomposition, combustion, single displacement or double displacement reaction. caco3àcao+co2
question 9
indicate whether the reaction is an example of a synthesis, decomposition, combustion, single displacement or double displacement reaction. p4+3o2à2p2o3
question 10
indicate whether the reaction is an example of a synthesis, decomposition, combustion, single displacement or double displacement reaction. c3h6o+4o2à3co2+3h2o
question 11
balance the following equation to satisfy the law of conservation of mass. nai + pb(so4)2 àpbi4+ na2so4 enter the coefficients in order from left to right, separated for a comma. if there is no coefficient, use a 1. example: h2+o2àh2o is balanced as 2h2+o2à2h2o enter the answer as 2,1,2 question 12
balance the following equation to satisfy the law of conservation of mass. fe(oh)3à fe2o3+ h2o enter the coefficients in order from left to right, separated for a comma. if there is no coefficient, use a 1. example: h2+o2àh2o is balanced as 2h2+o2à2h2o enter the answer as 2,1,2 question 13
5 points extra credit
balance the following equation to satisfy the law of conservation of mass. nh3+ o2à no+ h2o enter the coefficients in order from left to right, separated for a comma. if there is no coefficient, use a 1. example: h2+o2àh2o is balanced as 2h2+o2à2h2o enter the answer as 2,1,2 question 14
predict the products of the following reaction: nai+cacl2à separate the products with a comma. do not use subscripts. for example, if the products are nh3 and h2o the answer would be entered as: nh3, h2o
question 15
predict the products of the following reaction: li+ kno3à (lithium is above potassium on the activity series for metals) separate the products with a comma. do not use subscripts. for example, if the products are nh3 and h2o the answer would be entered as: nh3, h2o
question 16
5 points extra credit
predict the products of the following reaction: rbno2 +baco3à separate the products with a comma. do not use subscripts. for example, if the products are nh3 and h2o the answer would be entered as: nh3, h2o if needed for polyatomic ions, use parenthesis. example: al(no3)3 should be entered as al(no3)3
quick! i have 7 minutes left on the timer!

Answers: 3
Another question on Mathematics

Mathematics, 21.06.2019 21:30
(c) the diagram shows the first three shapes of a matchsticks arrangement.first shape = 3second shape = 5third shape = 7given m represents the number of matchstick used to arrange the n'th shape.(i) write a formula to represent the relation between m and n. express m as the subjectof the formula.(ii) 19 matchsticks are used to make the p'th shape. find the value of p.
Answers: 2

Mathematics, 21.06.2019 21:30
(03.03 mc) choose the graph below that correctly represents the equation 2x + 4y = 24. (5 points) select one: a. line through the points 0 comma 6 and 12 comma 0 b. line through the points 0 comma negative 6 and 12 comma 0 c. line through the points 0 comma negative 12 and 6 comma 0 d. line through the points 0 comma 12 and 6 comma 0
Answers: 1

Mathematics, 21.06.2019 21:30
In a test for esp (extrasensory perception), the experimenter looks at cards that are hidden from the subject. each card contains either a star, a circle, a wave, a cross or a square.(five shapes) as the experimenter looks at each of 20 cards in turn, the subject names the shape on the card. when the esp study described above discovers a subject whose performance appears to be better than guessing, the study continues at greater length. the experimenter looks at many cards bearing one of five shapes (star, square, circle, wave, and cross) in an order determined by random numbers. the subject cannot see the experimenter as he looks at each card in turn, in order to avoid any possible nonverbal clues. the answers of a subject who does not have esp should be independent observations, each with probability 1/5 of success. we record 1000 attempts. which of the following assumptions must be met in order to solve this problem? it's reasonable to assume normality 0.8(1000), 0.2(1000)%30 approximately normal 0.8(1000), 0.2(1000)% 10 approximately normal srs it is reasonable to assume the total number of cards is over 10,000 it is reasonable to assume the total number of cards is over 1000
Answers: 1

You know the right answer?
50 pts and bra8inliest if u get it right and answer all
question 1 options:
indi...
question 1 options:
indi...
Questions

Chemistry, 18.09.2019 14:50

History, 18.09.2019 14:50






Mathematics, 18.09.2019 14:50


History, 18.09.2019 14:50

Mathematics, 18.09.2019 14:50


Biology, 18.09.2019 14:50


Social Studies, 18.09.2019 14:50

Health, 18.09.2019 14:50

Mathematics, 18.09.2019 14:50

Mathematics, 18.09.2019 14:50


History, 18.09.2019 14:50

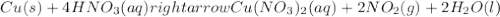 : oxidation reduction
: oxidation reduction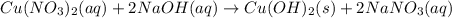 : precipitation
: precipitation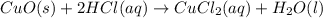 : Double displacement
: Double displacement

