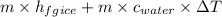Engineering, 09.12.2020 21:10 nev322
15 kg of ice at 0°C is heated to water of temperature 20°C. What is the amount of heat required if the specific latent heat of ice fusion is 335 kJ/kg and the specific heat capacity of water is 4.19kJ/kg/°C?

Answers: 3
Another question on Engineering

Engineering, 03.07.2019 23:20
Two technicians are discussing the intake air temperature (iat) sensor. technician a says that the computer uses the iat sensor as a backup to the engine coolant temperature (ect) sensor. technician b says that the powertrain control module (pcm) will subtract the calculated amount of fuel if the air measures hot. who is correct
Answers: 3

Engineering, 04.07.2019 18:10
Acompressor receives the shaft work to decrease the pressure of the fluid. a)- true b)- false
Answers: 3

Engineering, 04.07.2019 18:20
Inadequate stores control is not an obstacle to effective work order system. (clo4) a)-true b)-false
Answers: 3

Engineering, 04.07.2019 18:20
Athin walled concentric tube exchanger is used to cool engine oil from 160°c to 60°c with water that is available at 25°c acting as a coolant. the oil and water flow rates are each at 2 kg/s, and the diameter of the inner tube is 0.5 m and the corresponding value of the overall heat transfer coefficient is 250 w/m2. oc. how long must the heat exchanger be to accomplish the desired cooling? cpwater=4.187 kj/kg-candcpengine el=2.035 kj/kg·°c, oil . 120]
Answers: 1
You know the right answer?
15 kg of ice at 0°C is heated to water of temperature 20°C. What is the amount of heat required if...
Questions


Mathematics, 26.02.2021 23:50

Mathematics, 26.02.2021 23:50


Arts, 26.02.2021 23:50


History, 26.02.2021 23:50

Mathematics, 26.02.2021 23:50

Mathematics, 26.02.2021 23:50

Health, 26.02.2021 23:50



Physics, 26.02.2021 23:50

Biology, 26.02.2021 23:50

Mathematics, 26.02.2021 23:50





Mathematics, 26.02.2021 23:50

 = 335 kJ/kg
= 335 kJ/kg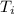 = 0° C
= 0° C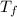 = 20° C
= 20° C