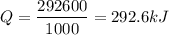Engineering, 30.03.2020 21:25 coolkitty35
Find the amount of energy (Q) required to raise the temperature of the water in kilo joules (KJ).
Given mass of water m = 2kg = 2000g.
Temperature difference ΔΤ = T2 – T1 = 60 °C – 25 °C = 35 °C .
Specific heat of water = 4.18 J/g* °C .
Therefore, Q =
kilo joules.

Answers: 3
Another question on Engineering

Engineering, 03.07.2019 15:10
Apiston-cylinder with a volume of 0.25 m3 holds 1 kg of air (r 0.287 k/kgk) at a temperature of 100 c. heat transfer to the cylinder causes an isothermal expansion of the piston until the volume triples. how much heat is added to the piston-cylinder?
Answers: 3

Engineering, 04.07.2019 18:10
Aloaded platform of total mass 500 kg is supported by a dashpot and by a set of springs of effective stiffness 72 kn/m. it is observed that when the platform is depressed through a distance x = 12.5 cm below its equilibrium position and then released without any initial velocity; it reaches its equilibrium position in the shortest possible time without overshoot. find the position and velocity of the loaded platform 0.10 sec. after its release. if a further load of 400 kg is added to the platform, find, i) the frequency of damped vibrations, and i) the amplitude of vibration after 2 complete oscillations, given that the initial amplitude is 15 cm.
Answers: 1

Engineering, 04.07.2019 18:10
Afluid flows with a velocity field given by v=(x/t)i.. determine the local and convective accelerations when x=3 and t=1.
Answers: 2

Engineering, 04.07.2019 18:10
Different types of steels contain different elements that alter the characteristics of the steel. for each of the following elements, explain what the element does when alloyed with steel.
Answers: 2
You know the right answer?
Find the amount of energy (Q) required to raise the temperature of the water in kilo joules (KJ).
Questions

History, 31.07.2019 18:30




Biology, 31.07.2019 18:30

Mathematics, 31.07.2019 18:30

History, 31.07.2019 18:30

Business, 31.07.2019 18:30



Mathematics, 31.07.2019 18:30


Social Studies, 31.07.2019 18:30

History, 31.07.2019 18:30

Mathematics, 31.07.2019 18:30

Mathematics, 31.07.2019 18:30

Mathematics, 31.07.2019 18:30

English, 31.07.2019 18:30


Chemistry, 31.07.2019 18:30