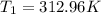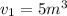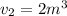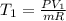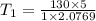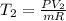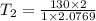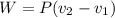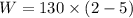Engineering, 14.02.2020 05:26 mari200150
The volume of 1 kg of helium in a piston–cylinder device is initially 5 m3. Now helium is compressed to 2 m3 while its pressure is maintained constant at 130 kPa. Determine the initial and final temperatures of helium as well as the work required to compress it, in kJ. The gas constant of helium is R = 2.0769 kJ/kg·K.

Answers: 1
Another question on Engineering

Engineering, 04.07.2019 18:10
What difference(s) did you notice using a pneumatic circuit over hydraulic circuit.explain why the pneumatic piston stumbles when it hits an obstacle.
Answers: 2

Engineering, 04.07.2019 18:10
Afluid flows with a velocity field given by v=(x/t)i.. determine the local and convective accelerations when x=3 and t=1.
Answers: 2

Engineering, 04.07.2019 18:10
Which of the following ziegler nichols tuning methods the response of the controller to a step input should exhibit an s-shaped curve? a)-open loop mode b)-closed loop mode c)-both modes (open & closed) d)-none of the modes (open & closed)
Answers: 3

Engineering, 04.07.2019 18:20
Determine the damped natural frequencies and the steady state response of a decoupled damped forced two degrees of freedom system. 10ä1 + 2q1 20q1 10 cos t; 10q2 +4q2 + 40q2 10 cos t
Answers: 3
You know the right answer?
The volume of 1 kg of helium in a piston–cylinder device is initially 5 m3. Now helium is compressed...
Questions

Mathematics, 23.09.2021 17:10


Advanced Placement (AP), 23.09.2021 17:10


Physics, 23.09.2021 17:20



Mathematics, 23.09.2021 17:20

Mathematics, 23.09.2021 17:20

Chemistry, 23.09.2021 17:20




Mathematics, 23.09.2021 17:20

Mathematics, 23.09.2021 17:20


Chemistry, 23.09.2021 17:20

Social Studies, 23.09.2021 17:20