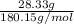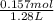Chemistry, 21.07.2019 15:00 lovedavis9544
Calculate the molarity of each solution. 28.33 g c6h12o6 in 1.28 l of solution

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 22.06.2019 06:00
One does not belong why? ice, gold ,wood ,diamond and table salt
Answers: 1

Chemistry, 22.06.2019 20:00
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2

You know the right answer?
Calculate the molarity of each solution. 28.33 g c6h12o6 in 1.28 l of solution...
Questions


History, 26.08.2020 14:01

English, 26.08.2020 14:01


Biology, 26.08.2020 14:01

Chemistry, 26.08.2020 14:01


Health, 26.08.2020 14:01

Mathematics, 26.08.2020 14:01


Mathematics, 26.08.2020 14:01



Mathematics, 26.08.2020 14:01


Mathematics, 26.08.2020 14:01

Mathematics, 26.08.2020 14:01

English, 26.08.2020 14:01

Spanish, 26.08.2020 14:01

Health, 26.08.2020 14:01

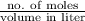
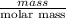
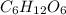 is 180.15 g/mol. Therefore, number of moles present will be as follows.
is 180.15 g/mol. Therefore, number of moles present will be as follows.