Chemistry, 21.07.2019 23:00 asuhdude57
)determine the theoretical yield and the percent yield if 21.8 g of k2co3 is produced from reacting 27.9 g ko2 with 29.0 l of co2 (at stp). the molar mass of ko2 = 71.10 g/mol and k2co3 = 138.21 g/mol.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:20
Sugar is dissolved in water. which is the solute? sugar neither both water
Answers: 1

Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 3

Chemistry, 22.06.2019 20:00
What is the molarity of the solution produced when 145 g of nacl is dissolved in sufficient water to prepare 2.75 l of solution?
Answers: 1

Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1
You know the right answer?
)determine the theoretical yield and the percent yield if 21.8 g of k2co3 is produced from reacting...
Questions


Mathematics, 17.12.2021 06:30

Social Studies, 17.12.2021 06:30

Mathematics, 17.12.2021 06:30


Mathematics, 17.12.2021 06:30


Mathematics, 17.12.2021 06:30


Mathematics, 17.12.2021 06:30

Mathematics, 17.12.2021 06:30

Mathematics, 17.12.2021 06:30


Mathematics, 17.12.2021 06:30



Mathematics, 17.12.2021 06:30


Chemistry, 17.12.2021 06:30

Mathematics, 17.12.2021 06:30

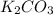 = 27.089 g
= 27.089 g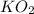 = 27.9 g
= 27.9 g
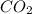 = 29.0 L (At STP)
= 29.0 L (At STP)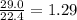 mole of
mole of 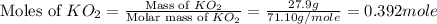
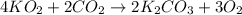
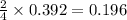 moles of
moles of