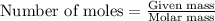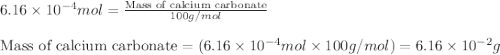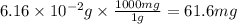Chemistry, 23.07.2019 10:00 coralstoner6793
Acid precipitation dripping on limestone produces carbon dioxide by the following reaction: caco3(s) + 2h+(aq) > ca(2+)(aq) + co2(g) + h2o (l) 15.5ml of co2 was produced at 25*c and 738.0 mmhg how man moles of co2 were produced? how many milligrams of caco3 were consumed?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:00
What is the molality of a solution that has 4 mol of kci in 0.800 kg of water
Answers: 3

Chemistry, 21.06.2019 21:50
H2so4(aq) + mg(s)—> mgso4(aq) +h2(g) which substance is the acid in the reaction?
Answers: 3

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 3

You know the right answer?
Acid precipitation dripping on limestone produces carbon dioxide by the following reaction: caco3(s...
Questions





Mathematics, 12.03.2020 18:08










Arts, 12.03.2020 18:09




Computers and Technology, 12.03.2020 18:10

Mathematics, 12.03.2020 18:10

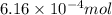 and the mass of calcium carbonate is 61.6 mg
and the mass of calcium carbonate is 61.6 mg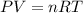
![25^oC=[25+273]K=298K](/tpl/images/0123/0054/df1f6.png)
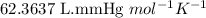

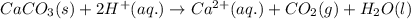
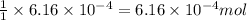 of calcium carbonate
of calcium carbonate