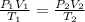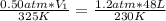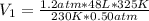Chemistry, 23.07.2019 10:30 carlydays3331
Ihave an unknown volume of gas at a pressure of 0.50 atm a temperature of 325 k. if i raise the pressure to 1.2 atm, decrease the temperature to 230 k, and measure the final volume to be 48 liters, what was the initial volume of the gas

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:00
What is the molarity of 60.0 grams of naoh dissolved in 750 milliliters of water? a) 1.1 m b) 2.0 m c) 12 m d) 75 m
Answers: 1

Chemistry, 22.06.2019 12:30
Which of the following describes a compound? (hint: carbon and oxygen bo a. a piece of pure carbon, containing only carbon atoms b. oxygen gas surrounding a solid piece of carbon c. a substance made of two oxygen atoms for each carbon atom carbon and oxygen atoms mixed without being bonded together
Answers: 1

Chemistry, 22.06.2019 13:00
What happens to the average kinetic energy of a gas when the particles of the gas collide against each other at a constant temperature and volume? explain your answer.
Answers: 3

Chemistry, 22.06.2019 14:50
How are evaporation and sublimation similar? a both involve the formation of a gas. b both release energy to the surroundings. c both take place throughout a solid. d both take place at the surface of a liquid.
Answers: 1
You know the right answer?
Ihave an unknown volume of gas at a pressure of 0.50 atm a temperature of 325 k. if i raise the pres...
Questions









Chemistry, 06.10.2019 07:30








Biology, 06.10.2019 07:30

Biology, 06.10.2019 07:30


History, 06.10.2019 07:30