Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 12:10
Building glycogen from glucose molecules is an example of
Answers: 3

Chemistry, 22.06.2019 23:00
How does the value of the equilibrium constant show that a reaction reaches equilibrium very quickly? (a) the equilibrium constant is large. (b) the equilibrium constant is small. (c) the equilibrium constant is zero. (d) the value of the equilibrium constant does not show how quickly a reaction comes to equilibrium.
Answers: 1
You know the right answer?
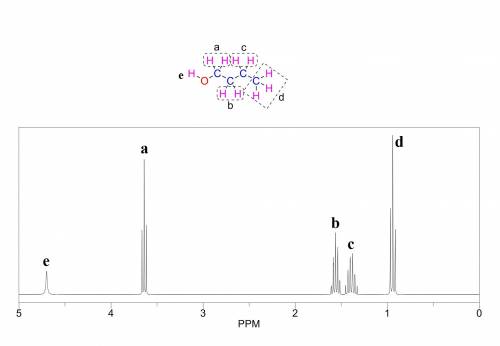
How many signals would you expect to find in the 1h nmr spectrum of 1 butanol?...
Questions

Mathematics, 15.11.2020 02:50

Mathematics, 15.11.2020 02:50


Biology, 15.11.2020 02:50

English, 15.11.2020 02:50

Advanced Placement (AP), 15.11.2020 02:50

History, 15.11.2020 02:50


History, 15.11.2020 02:50

Mathematics, 15.11.2020 02:50

Health, 15.11.2020 02:50

Geography, 15.11.2020 02:50



Geography, 15.11.2020 02:50

Mathematics, 15.11.2020 02:50

Computers and Technology, 15.11.2020 02:50

Mathematics, 15.11.2020 02:50