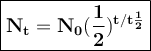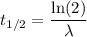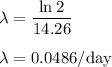Chemistry, 23.07.2019 21:00 jalenshayewilliams
The half-life of phosphorus-32 is 14.26 days. calculate its decay constant. express the decay constant numerically in inverse days.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 06:00
If you burn 10 kilograms of wood in a fire (combustion) what is the weight of the products after the fire has finished burning the wood?
Answers: 3

Chemistry, 22.06.2019 10:00
How many mmols of tris-hcl are there in 100 ml of a 100 mm tris-hcl buffer solution at ph 8.1? note that the 100 mm refers to the sum of tris and tris-hcl concentrations?
Answers: 3

Chemistry, 22.06.2019 12:00
Why are people not able to skip a dive to the deepest part of the ocean
Answers: 1
You know the right answer?
The half-life of phosphorus-32 is 14.26 days. calculate its decay constant. express the decay consta...
Questions




Social Studies, 18.07.2019 02:00


Social Studies, 18.07.2019 02:00

Social Studies, 18.07.2019 02:00

Social Studies, 18.07.2019 02:00




Social Studies, 18.07.2019 02:00

Social Studies, 18.07.2019 02:00

English, 18.07.2019 02:00


Chemistry, 18.07.2019 02:00

Chemistry, 18.07.2019 02:00

Mathematics, 18.07.2019 02:10