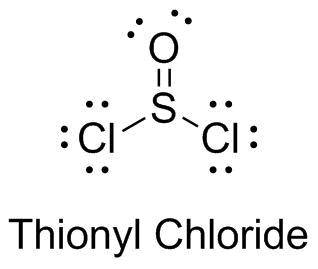
Chemistry, 24.07.2019 06:30 sofialarrea13
What noble gas has the same ground state electron configuration as the oxygen in thionyl chloride?

Answers: 1
Another question on Chemistry


Chemistry, 23.06.2019 06:00
In an exothermic reaction at equilibrium, what is the effect of lowering the temperature? a. the reaction makes more products. b. the reaction makes more reactants. c. the reaction is unchanged.
Answers: 1

Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1

Chemistry, 23.06.2019 11:50
Charles's law describes the relationship of the volume and temperature of gas at a constant mass and pressure. according to this law, what would happen to the temperature of the gas if its volume decreased from 1.0 l to 0.50 l?
Answers: 3
You know the right answer?
What noble gas has the same ground state electron configuration as the oxygen in thionyl chloride?...
Questions


Biology, 07.01.2021 19:20

English, 07.01.2021 19:20

Mathematics, 07.01.2021 19:20

Spanish, 07.01.2021 19:20

History, 07.01.2021 19:20


Geography, 07.01.2021 19:20



Mathematics, 07.01.2021 19:20

Health, 07.01.2021 19:20




Mathematics, 07.01.2021 19:20

English, 07.01.2021 19:20


Mathematics, 07.01.2021 19:20

English, 07.01.2021 19:20