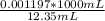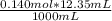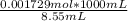Chemistry, 25.07.2019 07:00 hezekiahmharris
The initial volume of hcl was 1.25 ml and lioh was 2.65 ml. the final volume of hcl was 13.60 ml and lioh was 11.20 ml. if the lioh was .140 m what was the molarity of hcl ? if the same volumes were used from question 4, but the hcl was .140 m , what would the molarity of lioh be ?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:50
Working with si (metric) units for each of the following commonly used measurements, indicate its symbol. liter gram milliliter kilogram meter centigram milligram centimeter kilometer second millimeter milliseconds
Answers: 1

Chemistry, 22.06.2019 01:30
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 13:00
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2

Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3
You know the right answer?
The initial volume of hcl was 1.25 ml and lioh was 2.65 ml. the final volume of hcl was 13.60 ml and...
Questions

Social Studies, 13.02.2020 02:59


Biology, 13.02.2020 02:59



Medicine, 13.02.2020 02:59


Social Studies, 13.02.2020 02:59


Biology, 13.02.2020 02:59


History, 13.02.2020 02:59



Computers and Technology, 13.02.2020 02:59



History, 13.02.2020 02:59

Mathematics, 13.02.2020 02:59

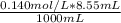 = 0.001197 mol
= 0.001197 mol