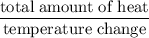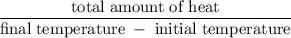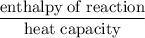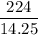Chemistry, 25.07.2019 14:00 wyattjefferds05
A6.55 g sample of aniline (c6h5nh2, molar mass = 93.13 g/mol) was combusted in a bomb calorimeter with a heat capacity of 14.25 kj/°c. if the initial temperature was 32.9°c, use the information below to determine the value of the final temperature of the calorimeter. 4 c6h5nh2(l) + 35 o2(g) → 24 co2(g) + 14 h2o(g) + 4 no2(g)

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Zinc + lead(ii) nitrate yield zinc nitrate + leadwhat's the chemical equation for this?
Answers: 1

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3


You know the right answer?
A6.55 g sample of aniline (c6h5nh2, molar mass = 93.13 g/mol) was combusted in a bomb calorimeter wi...
Questions

Mathematics, 07.12.2021 23:40


History, 07.12.2021 23:40

Mathematics, 07.12.2021 23:40

Mathematics, 07.12.2021 23:40



Mathematics, 07.12.2021 23:40


Mathematics, 07.12.2021 23:40

Mathematics, 07.12.2021 23:40


Mathematics, 07.12.2021 23:40


Mathematics, 07.12.2021 23:40


Health, 07.12.2021 23:40



Mathematics, 07.12.2021 23:40

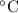 .
.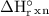 = 1.28
= 1.28 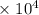 kJ.
kJ.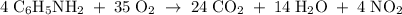
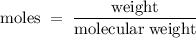
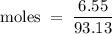
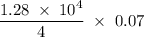
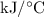 .
.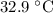 .
.