Chemistry, 25.07.2019 14:00 frenchpotaetoes2832
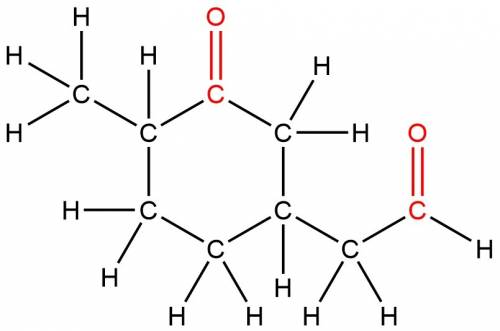
The molecule below contains both an aldehyde and a ketone functional group. select only the carbonyl carbon atom of the aldehyde functional group. identify the carbon atom by selecting the atom and assigning it a map number of 1. to do this, right-click on an atom and choose atom properties.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 23.06.2019 02:00
Why does ammonia, nh3, behave as a base when it reacts with an acid? z
Answers: 2

Chemistry, 23.06.2019 04:00
What are the names of these two interactions with cattle and how do they differ from each other
Answers: 3
You know the right answer?
The molecule below contains both an aldehyde and a ketone functional group. select only the carbonyl...
Questions

Mathematics, 14.10.2019 20:00

Mathematics, 14.10.2019 20:00




History, 14.10.2019 20:00


Mathematics, 14.10.2019 20:00

Mathematics, 14.10.2019 20:00




History, 14.10.2019 20:00



Mathematics, 14.10.2019 20:00

History, 14.10.2019 20:00