Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 02:00
For each of the following types of reactions, write a general reaction formula in the symbolic form—for example, a + b → ab. single-displacement double-displacement synthesis decomposition
Answers: 1

You know the right answer?
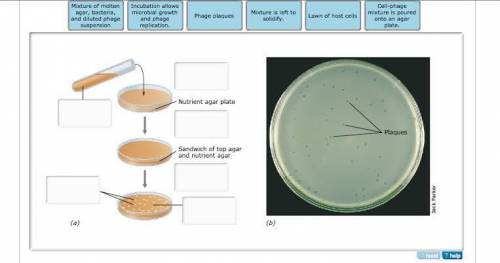
Correctly identify the steps and elements in this bacteriophage plaque assay...
Questions

Health, 28.11.2019 16:31

History, 28.11.2019 16:31


Health, 28.11.2019 16:31

Mathematics, 28.11.2019 16:31


Chemistry, 28.11.2019 16:31



Physics, 28.11.2019 16:31

Social Studies, 28.11.2019 16:31


Chemistry, 28.11.2019 16:31


History, 28.11.2019 16:31



World Languages, 28.11.2019 16:31

Advanced Placement (AP), 28.11.2019 16:31

Mathematics, 28.11.2019 16:31