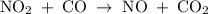The reaction between no2 and co to produce no and co2 is thought to occur in two steps: step 1: no2 + no2 no + no3 step 2: no3 + co no2 + co2 the experimental rate law is rate = k[no2]2. (a) write the equation for the overall reaction. do not include phase abbreviations. (b) identify the intermediate(s). no3 no2 co no co2

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 12:30
Select the correct answer. what is the nature of the se-cl bond in a molecule of selenium chloride (secl2) if the electronegativity value of selenium is 2.55 and that of chlorine is 3.16?
Answers: 3

Chemistry, 21.06.2019 19:30
Agas has a volume of 0.7 l at 300 mmhg. what would be the new volume at 900 mmhg
Answers: 1

Chemistry, 22.06.2019 09:00
How are isotopes of the same chemical element alike? how are they different?
Answers: 1

You know the right answer?
The reaction between no2 and co to produce no and co2 is thought to occur in two steps: step 1: no...
Questions

Chemistry, 24.11.2020 18:30


Mathematics, 24.11.2020 18:30



Mathematics, 24.11.2020 18:30





Mathematics, 24.11.2020 18:30

Mathematics, 24.11.2020 18:30

Mathematics, 24.11.2020 18:30

Mathematics, 24.11.2020 18:30


Mathematics, 24.11.2020 18:30

Mathematics, 24.11.2020 18:30

Health, 24.11.2020 18:30


Mathematics, 24.11.2020 18:30

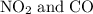 to produce NO and
to produce NO and 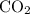 occurs in the given two steps.
occurs in the given two steps.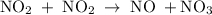

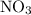 because it act as an intermediate and is utilized in the next step.
because it act as an intermediate and is utilized in the next step.