
Chemistry, 27.07.2019 17:30 nockturnal1993
In ammonia, a central nitrogen atom is bonded to three hydrogen atoms. similarly, boron trifluoride has a central boron atom bonded to three fluorine atoms. however, ammonia is pyramidal and boron trifluoride is trigonal planar in shape. which statement justifies this difference in their structure? the lone pair of electrons in boron trifluoride increases the bond angle between the bonding pairs, giving it a planar structure. the lone pair of electrons in boron trifluoride decreases the bond angle between the bonding pairs, giving it a planar structure. the lone pair of electrons in boron trifluoride stabilizes the bond angle between the bonding pairs, giving it a planar structure. the lone pair of electrons in ammonia increases the bond angle between the bonding pairs, giving it a pyramidal structure. the lone pair of electrons on the nitrogen in ammonia decreases the bond angle between the bonding pairs, giving it a pyramidal structure.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 3

Chemistry, 22.06.2019 21:00
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1


You know the right answer?
In ammonia, a central nitrogen atom is bonded to three hydrogen atoms. similarly, boron trifluoride...
Questions



Mathematics, 27.02.2020 00:28


Social Studies, 27.02.2020 00:28








Computers and Technology, 27.02.2020 00:28







Computers and Technology, 27.02.2020 00:28

![:{\text{Number of electrons}} =\frac{1}{2}[V+N-C+A]](/tpl/images/0139/5742/08b6a.png)
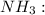
![{\text{Number of electrons}} =\frac{1}{2}[5+3-0+0]=4](/tpl/images/0139/5742/46c6e.png)
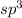 and the electronic geometry of the molecule will be tetrahedral.
and the electronic geometry of the molecule will be tetrahedral.
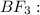
![{\text{Number of electrons}} =\frac{1}{2}[3+3-0+0]=3](/tpl/images/0139/5742/beb45.png)
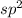 and the electronic geometry of the molecule will be trigonal planar.
and the electronic geometry of the molecule will be trigonal planar.



