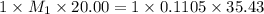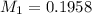You are given a solution of hcooh (formic acid) with an approximate concentration of 0.20 m and you will titrate this with a 0.1105 m naoh. you add 20.00 ml of hcooh to the beaker before titrating, and it requires 35.43 ml of naoh to reach the end point. what is the concentration of the hcooh solution?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
The balanced chemical equation for this lab is: 3cucl2(aq) + 2al(s) 3cu(s) + 2alcl3(aq) if 10.5 g copper chloride react with 12.4 g aluminum, what is the limiting reactant?
Answers: 3

Chemistry, 22.06.2019 06:40
Three alkali metals in group 1 are a. calcium, strontium, barium b. boron, aluminum, gallium c. sodium, potassium, rubidium d. fluorine, iodine, chlorine
Answers: 1

Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2

You know the right answer?
You are given a solution of hcooh (formic acid) with an approximate concentration of 0.20 m and you...
Questions

Mathematics, 05.05.2020 08:47



Mathematics, 05.05.2020 08:47


Mathematics, 05.05.2020 08:47



Mathematics, 05.05.2020 08:47

Health, 05.05.2020 08:47

Mathematics, 05.05.2020 08:47

Arts, 05.05.2020 08:47





Mathematics, 05.05.2020 08:47

Health, 05.05.2020 08:47

English, 05.05.2020 08:47

Mathematics, 05.05.2020 08:47


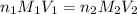
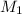 = molarity of
= molarity of 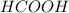 solution = ?
solution = ?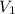 = volume of
= volume of 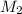 = molarity of
= molarity of 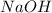 solution = 0.1105 M
solution = 0.1105 M
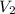 = volume of
= volume of 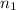 = valency of
= valency of 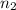 = valency of
= valency of