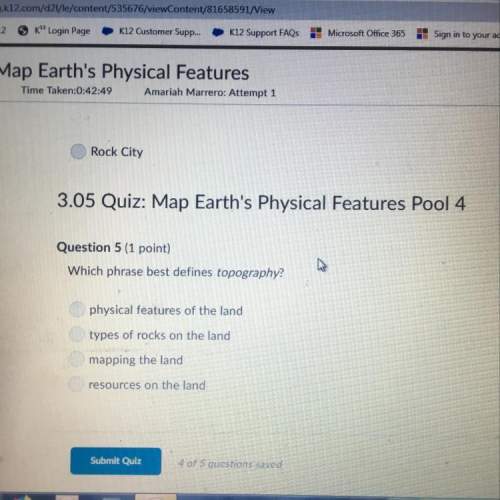
Chemistry, 29.07.2019 01:30 luljrjr4130
A25.0 ml sample of 0.150 m butanoic acid is titrated with a 0.150 m naoh solution. what is the ph after 26.0 ml of base is added? the ka of butanoic acid is 1.5 ⋅ 10-5. a 25.0 ml sample of 0.150 m butanoic acid is titrated with a 0.150 m naoh solution. what is the ph after 26.0 ml of base is added? the ka of butanoic acid is 1.5 10-5. 4.84 2.54 7.00 4.81 11.47

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Two atoms interact with each other as shown by the equation. complete the equation by filling in the missing parts. 1 2 3 4 5 h he li
Answers: 2

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 04:00
Write the empirical chemical formula of calcium with a mass percent of 38.8, phosphorus with a mass percent of 20.0, and oxygen with a mass percent of 41.3.
Answers: 1

Chemistry, 22.06.2019 15:30
Which statement names the physical property of wood a. wood is softer than coal b. wood does not rust c. wood can rot d. wood can burn
Answers: 1
You know the right answer?
A25.0 ml sample of 0.150 m butanoic acid is titrated with a 0.150 m naoh solution. what is the ph af...
Questions




Mathematics, 10.12.2021 01:40

Biology, 10.12.2021 01:40


Mathematics, 10.12.2021 01:40


Mathematics, 10.12.2021 01:40


Mathematics, 10.12.2021 01:40

French, 10.12.2021 01:40

Mathematics, 10.12.2021 01:40

Mathematics, 10.12.2021 01:40


Arts, 10.12.2021 01:40



Spanish, 10.12.2021 01:40