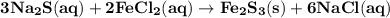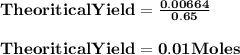Chemistry, 29.07.2019 09:00 josephaciaful
How many milliliters of 0.200 m fecl3 are needed to react with an excess of na2s to produce 1.38 g of fe2s3 if the percent yield for the reaction is 65.0%? 3 na2s(aq) + 2 fecl3(aq) → fe2s3(s) + 6 nacl(aq)?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 11:40
Which type of precipitation would most likely form when the surface air temperature is slightly below freezing and the air temperature increases as you move upward away from the ground?
Answers: 2

Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2

You know the right answer?
How many milliliters of 0.200 m fecl3 are needed to react with an excess of na2s to produce 1.38 g o...
Questions

Business, 16.02.2021 01:00






German, 16.02.2021 01:00

Biology, 16.02.2021 01:00

English, 16.02.2021 01:00

Chemistry, 16.02.2021 01:00


Mathematics, 16.02.2021 01:00





History, 16.02.2021 01:00


Mathematics, 16.02.2021 01:00

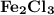 is needed to react with an excess of
is needed to react with an excess of 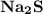 to produce 1.38 g of
to produce 1.38 g of 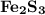 .
.