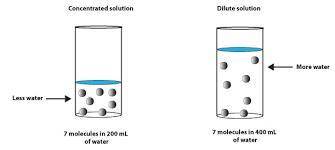
Chemistry, 29.07.2019 14:00 jasontbyrer
Which of the following solution is more dilute and explain why? a)1m b)2m c)0.1m or d)0.009m

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Will mark brainliest 26. which of these statements are true? (3 points) a. gases are compressible b. gases fill their containers completely c. the pressure of a gas is independent of the temperature d. gases have mass e. gases exert pressure f. the pressure of a gas is dependent on the volume g. gas pressure results from the collisions between gas particles h. gases have a definite volume and shape
Answers: 1


Chemistry, 23.06.2019 07:30
In a laboratory determination of the atomic weight of tin, a sample of tin is weighed in a crucible. nitric acid is added, and the reaction proceeds to give a hydrated tin(iv)oxide plus no2and h2o. the hydrated tin(iv)oxide is then heated strongly and reacts as follows: sno2.xh2o(s)sno2(s)+ xh2o(g)the sno2is finally cooled and weighed in the crucible. explain the effect on the calculated atomic weight of tin that would result from each of the following experimental errors: (a)considerable spattering occurs when the nitric acid is added to the tin.(b)the hydrated tin(iv)oxide is not heated sufficiently to change it completely to tin oxide.
Answers: 2

Chemistry, 23.06.2019 13:00
Which of the following statements is true about both nuclear fusion and nuclear fission? they occur in the sun. heavy atoms are split. two light nuclei combine. some mass changes into energy.
Answers: 1
You know the right answer?
Which of the following solution is more dilute and explain why? a)1m b)2m c)0.1m or d)0.009m...
Questions



English, 03.10.2019 02:00

English, 03.10.2019 02:00

Mathematics, 03.10.2019 02:00

Mathematics, 03.10.2019 02:00


Mathematics, 03.10.2019 02:00

Mathematics, 03.10.2019 02:00


Social Studies, 03.10.2019 02:00

Mathematics, 03.10.2019 02:00



History, 03.10.2019 02:00



Mathematics, 03.10.2019 02:00

English, 03.10.2019 02:00