

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Identify the electromagnets with poles that are reversed from the electromagnet shown above
Answers: 3

Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1

Chemistry, 22.06.2019 19:50
When the mercury level in a barometer decreases that atmospheric pressure has
Answers: 3

Chemistry, 22.06.2019 21:20
One way in which the useful metal copper is produced is by dissolving the mineral azurite, which contains copper(ii) carbonate, in concentrated sulfuric acid. the sulfuric acid reacts with the copper(ii) carbonate to produce a blue solution of copper(ii) sulfate. scrap iron is then added to this solution, and pure copper metal precipitates out because of the following chemical reaction: (s) (aq) (s) (aq) suppose an industrial quality-control chemist analyzes a sample from a copper processing plant in the following way. he adds powdered iron to a copper(ii) sulfate sample from the plant until no more copper will precipitate. he then washes, dries, and weighs the precipitate, and finds that it has a mass of .
Answers: 2
You know the right answer?
A0.50 m solution of formic acid, hcooh, has a ph of 2.02. calculate the percent ionization of hcooh...
Questions


Mathematics, 20.05.2020 11:57



English, 20.05.2020 11:57

Social Studies, 20.05.2020 11:57

Mathematics, 20.05.2020 11:57


English, 20.05.2020 11:57

Mathematics, 20.05.2020 11:57



Biology, 20.05.2020 11:57

Social Studies, 20.05.2020 11:57

Health, 20.05.2020 11:57


Biology, 20.05.2020 11:57

Mathematics, 20.05.2020 11:57

Mathematics, 20.05.2020 11:57

History, 20.05.2020 11:57

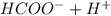
![[HCOO^{-}]_{eq} X [H^{+}]_{eq}](/tpl/images/0239/0647/2ee4e.png) )/
)/ ![[HCOOH]_{eq}](/tpl/images/0239/0647/dbf45.png)
 will be the pH of the solution.
will be the pH of the solution. ![[H^{+}]_{eq}](/tpl/images/0239/0647/0177f.png) =
= 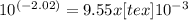 M
M  and
and 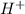 .
. ![[H^{+}]](/tpl/images/0239/0647/cab36.png) (M)
(M) ![[HCOO^{-}]](/tpl/images/0239/0647/03a1f.png) (M)
(M)  comes from the acid, and none from water.
comes from the acid, and none from water.  which is much higher than the 1.0 x
which is much higher than the 1.0 x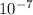 M [tex[H^{+}" alt=" 10^{-3}" /> M
M [tex[H^{+}" alt=" 10^{-3}" /> M  .
.  /0.50= 1.82 x
/0.50= 1.82 x 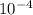
![[H^{+}]_{eq}](/tpl/images/0239/0647/0123b.png) x 100 /
x 100 /![[HCOOH]_{i}](/tpl/images/0239/0647/cd293.png)
 / (0.50)}x 100 = 1.91 %
/ (0.50)}x 100 = 1.91 %

