Chemistry, 30.07.2019 13:00 chantelljenkins2
Assume that 1.0 mol of c4h10 is completely burned in excess oxygen to form carbon dioxide and water. how many moles of co2 would be produced?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:40
In the reading, yao chen-yuan describes traveling to deliver a message. why was he willing to risk danger to travelto tientsin? he wanted to the boxers with their cause
Answers: 2

Chemistry, 22.06.2019 04:30
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 05:00
As you watch a surfer ride a wave towards the shoreline, what is the shoreline? a) displacement reference b) reference point c) coordinate plane d) cartesian boundary
Answers: 1

Chemistry, 22.06.2019 20:00
Listenbase your answer to the question on the information below.nuclear radiation is harmful to living cells, particularly to fast-growing cells, such as cancer cells and blood cells. an external beam of the radiation emitted from a radioisotope can be directed on a small area of a person to destroy cancer cells within the body.cobalt-60 is an artificially produced radioisotope that emits gamma rays and beta particles. one hospital keeps a 100.0-gram sample of cobalt-60 in an appropriate, secure storage container for future cancer treatment.which choice represents the correct product for the beta decay of the co-60? fe-60ni-60fe-61ni-61
Answers: 2
You know the right answer?
Assume that 1.0 mol of c4h10 is completely burned in excess oxygen to form carbon dioxide and water....
Questions

English, 11.03.2021 19:20

Mathematics, 11.03.2021 19:20

Mathematics, 11.03.2021 19:20


Business, 11.03.2021 19:20

History, 11.03.2021 19:20






History, 11.03.2021 19:20


Mathematics, 11.03.2021 19:20



Mathematics, 11.03.2021 19:20



Mathematics, 11.03.2021 19:20

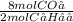 ) = 4 moles of CO₂
) = 4 moles of CO₂