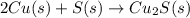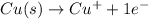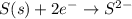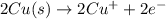Chemistry, 31.07.2019 06:30 Manuelperez1373
In the balanced redox reaction: 2 cu(s) + s(s) ® cu2s(s), how many electrons are gained or lost by each copper atom? select one: a. each copper atom gains two (2) electrons. b. each copper atom gains one (1) electron. c. each copper atom loses one (1) electron. d. each copper atom loses two (2) electrons.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:30
In an energy pyramid, which level has the most available energy?
Answers: 1

Chemistry, 22.06.2019 20:20
Which symbol can be used to indicate the pressure at which a chemical reaction is carried out? 25°c 2 atm pa
Answers: 2


Chemistry, 23.06.2019 00:00
#7 how does the structure of amino acids allow them to form a polypeptide? each amino acid has an amino group and a carboxyl group. each amino acid has a hydrogen atom and a carboxyl group. each amino acid has a carboxyl group and an r group. each amino acid has an r group and a hydrogen atom.
Answers: 1
You know the right answer?
In the balanced redox reaction: 2 cu(s) + s(s) ® cu2s(s), how many electrons are gained or lost by...
Questions



Computers and Technology, 13.12.2019 18:31








Computers and Technology, 13.12.2019 18:31