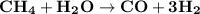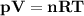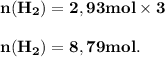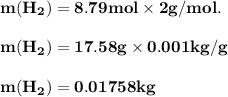Chemistry, 31.07.2019 18:30 katlynnschmolke
The reform reaction between steam and gaseous methane ( ch4 ) produces "synthesis gas," a mixture of carbon monoxide gas and dihydrogen gas. synthesis gas is one of the most widely used industrial chemicals, and is the major industrial source of hydrogen. suppose a chemical engineer studying a new catalyst for the reform reaction finds that 159. liters per second of methane are consumed when the reaction is run at 294.°c and 0.86atm . calculate the rate at which dihydrogen is being produced. give your answer in kilograms per second. be sure your answer has the correct number of significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:40
If equal masses of the listed metals were collected , which would have a greatest volume ? a. aluminum 2.70,b.zinc7.14,c.copper 8.92,d.lead 11.34
Answers: 2


Chemistry, 22.06.2019 15:00
Which substance is a steroid? cholesterol fatty acid monosaccharide trans fat
Answers: 1

Chemistry, 22.06.2019 17:00
What is the approximate vapor pressure when the gas condenses at 70 degrees celsius
Answers: 2
You know the right answer?
The reform reaction between steam and gaseous methane ( ch4 ) produces "synthesis gas," a mixture of...
Questions



Social Studies, 10.06.2021 22:40


Mathematics, 10.06.2021 22:40

Mathematics, 10.06.2021 22:40


History, 10.06.2021 22:40

SAT, 10.06.2021 22:40

Mathematics, 10.06.2021 22:40

Mathematics, 10.06.2021 22:40

Mathematics, 10.06.2021 22:40

Mathematics, 10.06.2021 22:40


Mathematics, 10.06.2021 22:40