Chemistry, 01.08.2019 09:00 tshort2000oyreb1
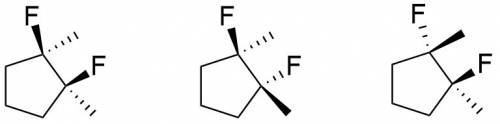
Draw every stereoisomer for 1,2-difluoro-1,2-dimethylcyclopenta ne. use wedge-and-dash bonds for the substituent groups, and be sure that they are drawn on the outside of the ring, adjacent to each other. the skeletal structure of one molecule is included to indicate the proper format; modify the x\'s on the given structure to create the first stereoisomer.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:40
How many liters of hydrogen gas will be produced at stp from the reaction of 7.179×10^23 atoms of magnesium with 54.219g of phosphoric acid (h3po4) the equation is 3mg + 2h3(> mg(po4)2+3h2
Answers: 1

Chemistry, 22.06.2019 05:30
Match the following vocabulary terms to their definitions. 1. amount of energy required to change 1 gram of material from the solid to the liquid state at its melting point 2. a measure of the kinetic energy of the particles of a substance 3. the amount of heat energy required to raise the temperature of 1 gram of liquid water from 14.5°c to 15.5°c 4. amount of energy required to change 1 gram of material from the liquid to the gaseous state at its boiling point 5. the amount of energy required to change 1 gram of a substance 1°c a. temperature b. latent heat of vaporization c. latent heat of fusion d. calorie e. specific heat
Answers: 1

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3
You know the right answer?
Draw every stereoisomer for 1,2-difluoro-1,2-dimethylcyclopenta ne. use wedge-and-dash bonds for the...
Questions





Mathematics, 14.06.2020 19:57

Mathematics, 14.06.2020 19:57


Health, 14.06.2020 19:57

Social Studies, 14.06.2020 19:57

Mathematics, 14.06.2020 19:57

Mathematics, 14.06.2020 19:57

Mathematics, 14.06.2020 19:57

Mathematics, 14.06.2020 19:57

Mathematics, 14.06.2020 19:57

Geography, 14.06.2020 19:57

History, 14.06.2020 19:57



Mathematics, 14.06.2020 19:57