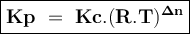Chemistry, 01.08.2019 16:30 afloareiandrei8615
An equilibrium mixture contains 0.750 mol of each of the products (carbon dioxide and hydrogen gas) and 0.200 mol of each of the reactants (carbon monoxide and water vapor) in a 1.00 l container. co(g)+h2o(g)↽−−⇀co2(g)+h2(g) how many moles of carbon dioxide would have to be added at constant temperature and volume to increase the amount of carbon monoxide to 0.300 mol once equilibrium has been reestablished?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:20
Which of these statements explains the difference between nuclear binding energy and the strong nuclear force ?
Answers: 3

Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1


Chemistry, 22.06.2019 23:30
Substance a is a nonpolar liquid and has only dispersion forces among its constituent particles. substance b is also a nonpolar liquid and has about the same magnitude of dispersion forces among its constituent particles. when substance a and b are combined, they spontaneously mix.
Answers: 1
You know the right answer?
An equilibrium mixture contains 0.750 mol of each of the products (carbon dioxide and hydrogen gas)...
Questions


Computers and Technology, 19.07.2019 23:00


Mathematics, 19.07.2019 23:00

History, 19.07.2019 23:00


Mathematics, 19.07.2019 23:00



Mathematics, 19.07.2019 23:00



Mathematics, 19.07.2019 23:00

Biology, 19.07.2019 23:00


Biology, 19.07.2019 23:00

Business, 19.07.2019 23:00

Geography, 19.07.2019 23:00


History, 19.07.2019 23:00

![\large {\boxed {\bold {Kc ~ = ~ \frac {[C] ^ m [D] ^ n} {[A] ^ p [B] ^ q}}}}](/tpl/images/0158/5375/9d1db.png)
![\large {\boxed {\bold {Kp ~ = ~ \frac {[pC] ^ m [pD] ^ n} {[pA] ^ p [pB] ^ q}}}}](/tpl/images/0158/5375/b3cf6.png)