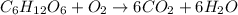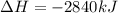Chemistry, 01.08.2019 16:30 carlalopezelox2244
The combustion of glucose, c6 h12 o6 (s), produces carbon dioxide, co2 (g), and water, h2 o(g), according to the equation below. mc020-1.jpg the enthalpy of the reaction is –2,840 kj. what is the heat of combustion, per mole, of glucose? –2,840 kj/mol –473.3 kj/mol 473.3 kj/mol 2,840 kj/mol

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2


Chemistry, 22.06.2019 16:30
Correct relationship between molecular formula and empirical formula
Answers: 1

Chemistry, 23.06.2019 00:00
(04.05 hc) analyze the given diagram of the carbon cycle below. part 1: which compound does c represent? part 2: name a process that could release this compound into the air. part 3: explain how the elements that form it are conserved during the carbon cycle. use complete sentences to explain your answer. justify how this compound was created from a recycling of carbon in the carbon cycle. use complete sentences to explain your answer.
Answers: 3
You know the right answer?
The combustion of glucose, c6 h12 o6 (s), produces carbon dioxide, co2 (g), and water, h2 o(g), acco...
Questions



Mathematics, 13.10.2019 00:50



Social Studies, 13.10.2019 00:50

Mathematics, 13.10.2019 00:50


Biology, 13.10.2019 00:50

Biology, 13.10.2019 00:50


Chemistry, 13.10.2019 00:50


History, 13.10.2019 00:50

Mathematics, 13.10.2019 00:50


Mathematics, 13.10.2019 00:50

Social Studies, 13.10.2019 00:50