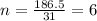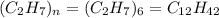Chemistry, 02.08.2019 19:30 tomuchsaucecj
What is the molecular formula of a compound given the molar mass of the compound is 186.5 gram and the empirical formula is c2h7?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 10:00
How many mmols of tris-hcl are there in 100 ml of a 100 mm tris-hcl buffer solution at ph 8.1? note that the 100 mm refers to the sum of tris and tris-hcl concentrations?
Answers: 3

Chemistry, 22.06.2019 16:00
No copying 15 pts how does a free-body diagram tell you about the net force on an object?
Answers: 2

Chemistry, 22.06.2019 23:00
What is the number of neutrons in an atom with atomic mass of 35
Answers: 2
You know the right answer?
What is the molecular formula of a compound given the molar mass of the compound is 186.5 gram and t...
Questions

Mathematics, 10.12.2020 22:50



Social Studies, 10.12.2020 22:50

Mathematics, 10.12.2020 22:50

Mathematics, 10.12.2020 22:50

English, 10.12.2020 22:50



Social Studies, 10.12.2020 22:50

Biology, 10.12.2020 22:50



World Languages, 10.12.2020 22:50



Mathematics, 10.12.2020 22:50


Geography, 10.12.2020 22:50

Mathematics, 10.12.2020 22:50

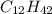
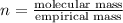
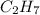 and the molar mass of compound is, 186.5 gram/mol.
and the molar mass of compound is, 186.5 gram/mol.