
Chemistry, 04.08.2019 05:00 mrsjenrucker
What is the molarity (m) of a solution prepared by dissolving 15.0 grams of na3po4 in enough water to make 250.0 ml of solution?

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 3

Chemistry, 23.06.2019 02:50
For questions 1 and 2, consider the following experimental data.hydrogen emission lines were detected at the following wavelengths (in nm): 121.6102.697.395.093.8question 1use the electromagnetic radiation classifications below and figure 1-1 in the introductory information for this lab (in the lab manual) to determine the nf value for the experimental data provided? wavelength, ? (nm) 650 700 550 600 400 450 500 visible spectrum wavelength, ? (m) 11 10 3 10 10 10 8 10 5 10 10 -10 10 9 10 10 10 10 -12 10 microwave radio infrared x-ray ultraviolet gamma 1020 1019 1018 1 1016 015 1014 01 12 109108 frequency, v (hz)a.1b. 2c. 3d. 4e. 5question 2using the data for the emission line with the longest wavelength, the known value of nf (from question 1 in this prelab), and the value of ni (deduced from the ? and nf values) calculate the rydberg constant for hydrogen (rh) in units of m-1.a) 1.097 x 10-11 m-1b) 5.921 x 107 m-1c) 1.097 x 10-2 m-1d) 9.252 x 106 m-1e) 1.097 x 107 m-1
Answers: 3

Chemistry, 23.06.2019 08:50
Reacting masses1 calcium carbonate breaks down on heating to produce calcium oxide and carbondioxide gas.caco3 + cao + co2a student heats 15 g of calcium carbonate strongly in a crucible.relative atomic masses (a): ca = 40, c = 12, o = 16.calculate the mass of calcium oxide produced by this reaction.(5 marks)
Answers: 3
You know the right answer?
What is the molarity (m) of a solution prepared by dissolving 15.0 grams of na3po4 in enough water t...
Questions





Chemistry, 26.10.2020 21:40


Biology, 26.10.2020 21:40

Social Studies, 26.10.2020 21:40


History, 26.10.2020 21:40

Health, 26.10.2020 21:40



Computers and Technology, 26.10.2020 21:40

English, 26.10.2020 21:40



Spanish, 26.10.2020 21:40

Physics, 26.10.2020 21:40

English, 26.10.2020 21:40

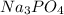 (solute) = 15 g
(solute) = 15 g![Molarity=\frac{\text{Moles of solute}\times 1000}{\text{Molar mass of solute}\times \text{volume of solution}}[tex]Now put all the given values in this formula, we get the molarity of the solution.[tex]Molarity=\frac{15g\times 1000}{163.94g/mole\times 250ml}=0.365mole/L](/tpl/images/0168/2467/caeda.png)


