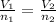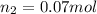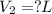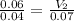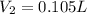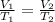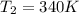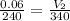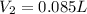Chemistry, 04.08.2019 06:50 dchirunga23
For this exercise, you can simulate the described conditions by changing the values in the run experiment tool of the simulation. to be able to measure the effects on the gas volume, slide the rspd bar in the properties box to v (l) so that the volume bar turns yellow. this way, volume becomes the only dependent variable. note that when volume is selected with rspd, its value can no longer be directly controlled. suppose a piston automatically adjusts to maintain a gas at a constant pressure of 14.20 atm . for the initial conditions, there are 0.04 mol of helium at a temperature of 240.00 k . this gas occupies a volume of 0.06 l under those conditions. what volume will the gas occupy if the number of moles is increased to 0.07 mol (n2) from the initial conditions? what volume will the gas occupy if the temperature is increased to 340.00 k (t2) from the initial conditions?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 12:00
What is a possible quantum number set for an electron in the 3s orbital of a magnesium atom
Answers: 1

Chemistry, 22.06.2019 17:00
The arrangement of particles is most ordered in a sample of
Answers: 1
You know the right answer?
For this exercise, you can simulate the described conditions by changing the values in the run exper...
Questions

Social Studies, 13.11.2020 16:20



Health, 13.11.2020 16:20

Spanish, 13.11.2020 16:20















Chemistry, 13.11.2020 16:20