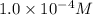Chemistry, 04.08.2019 03:50 elenaabad18
The hydrogen ion concentration of a vinegar solution is 0.00010 m. how is this concentration written in scientific notation? 1.0 mc014-1.jpg 10–5 m 1.0 mc014-2.jpg 10–4 m 1.0 mc014-3.jpg 104 m 1.0 mc014-4.jpg 105 m

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:30
It takes 945.kj/mol to break a nitrogen-nitrogen triple bond. calculate the maximum wavelength of light for which a nitrogen-nitrogen triple bond could be broken by absorbing a single photon.
Answers: 3

Chemistry, 21.06.2019 23:00
Why are the trends and exceptions to the trends in ionization energy observed?
Answers: 1

Chemistry, 22.06.2019 05:30
Liv sheldon given the balanced equation for an organic reaction: c2h2 + 2cl2 → c2h2cl4 this reaction is best classified as *
Answers: 1

Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1
You know the right answer?
The hydrogen ion concentration of a vinegar solution is 0.00010 m. how is this concentration written...
Questions

Physics, 13.01.2021 17:00

Physics, 13.01.2021 17:00


Chemistry, 13.01.2021 17:00

English, 13.01.2021 17:00

History, 13.01.2021 17:00

Social Studies, 13.01.2021 17:00

Arts, 13.01.2021 17:00


Mathematics, 13.01.2021 17:00


History, 13.01.2021 17:00


Mathematics, 13.01.2021 17:00

Mathematics, 13.01.2021 17:00

Mathematics, 13.01.2021 17:00


Mathematics, 13.01.2021 17:00