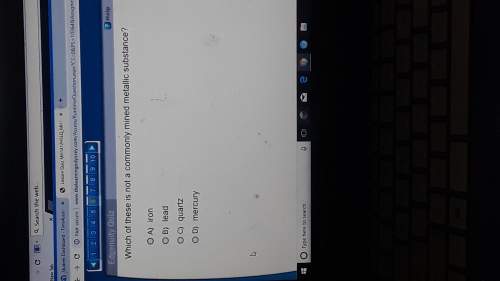
In an oxidation-reduction reaction, the number of electrons lost is
(1) equal to the number of...

Chemistry, 21.08.2019 07:50 autumnskye1
In an oxidation-reduction reaction, the number of electrons lost is
(1) equal to the number of electrons gained
(2) equal to the number of protons gained
(3) less than the number of electrons gained
(4) less than the number or protons gained

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Temperature and kinetic energy are proportional. a) adirectly b) directly c) indirectly
Answers: 2


Chemistry, 22.06.2019 04:30
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 06:30
Summarize possible ways in which phases of matter could combine to form a solution.
Answers: 2
You know the right answer?
Questions



Advanced Placement (AP), 23.06.2019 12:00



Spanish, 23.06.2019 12:00




Mathematics, 23.06.2019 12:00

Mathematics, 23.06.2019 12:00

Chemistry, 23.06.2019 12:00




Mathematics, 23.06.2019 12:00

Mathematics, 23.06.2019 12:00

Mathematics, 23.06.2019 12:00


Mathematics, 23.06.2019 12:00