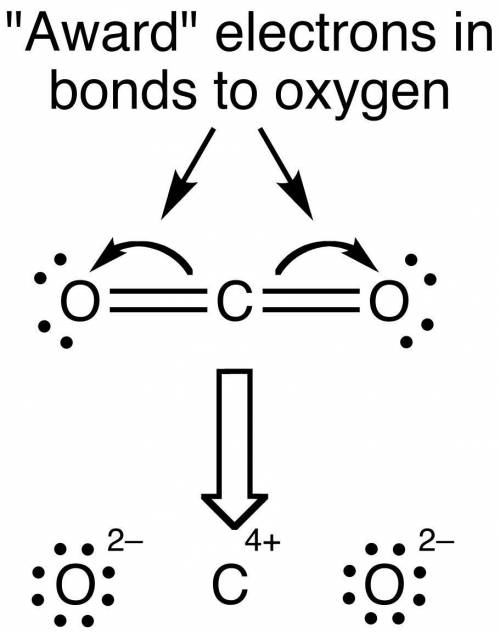
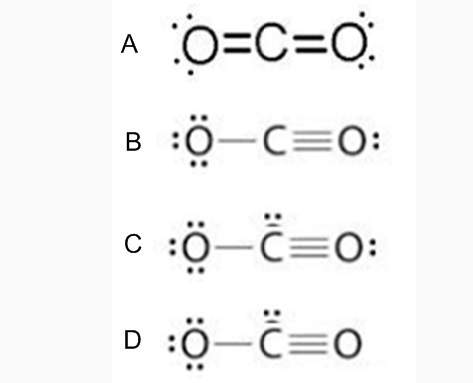
Of the options given, the correct one for the Lewis structure of CO₂ is option A.
In the covalent structure of CO₂, each atom will tend to have 8 electrons on its side, in order to comply with the octet rule. In this rule, an atom that has 8 electrons in its valence shell, will gain stability for having a noble gas electronic configuration.
Hence, the correct lewis structure for CO₂ is option A. In this configuration, each atom has 8 electrons on its side and all of them are neutral (the total charge of the molecule is zero). They all achieve the octet rule.
Although in option B all the atoms have 8 electrons (thus complying with the octet rule), in this configuration the oxygen atom on the left has a negative charge (-1) and the oxygen on the right side has a positive charge (+1). The total charge on the molecule is zero (it is neutral). In fact, this is a resonance structure of configuration A.
In option C, the total charge of the molecule is -2, so this is not a stable configuration. The number of electrons on carbon is wrong.
Finally, in option D the oxygen on the right side does not achieve the octet rule (it only has 6 electrons shared), and also the carbon atom has a charge of -2, and the oxygens have charges of -1 (left) and +3 (right), so this is not a stable configuration.
Therefore, the correct lewis structure for CO₂ is option A.
To find more about Lewis structure, go here:
I hope it helps you!