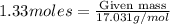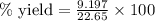Chemistry, 01.08.2019 21:30 05leslun42715
Ammonia (nh3) can be produced by the reaction of hydrogen gas with nitrogen gas: 3h2 + n2 = 2nh3 a chemist reacts 2.00 mol h2 with excess n2. the reaction yields 0.54 mol nh3. what is the percent yield of the reaction? a) 25% b) 40% c) 60% d) 80%

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 18:10
Measurements that have similar values are: a. usually accurate b. sometimes accurate c. always accurate d. never accurate
Answers: 1

Chemistry, 22.06.2019 23:30
The sum of the oxidation numbers in a neutral compound is always
Answers: 2


You know the right answer?
Ammonia (nh3) can be produced by the reaction of hydrogen gas with nitrogen gas: 3h2 + n2 = 2nh3 a...
Questions








Mathematics, 22.05.2020 22:02

Mathematics, 22.05.2020 22:02



Geography, 22.05.2020 22:02







Mathematics, 22.05.2020 22:02

Mathematics, 22.05.2020 22:02

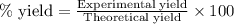 ....(1)
....(1)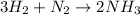
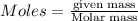 ....(2)
....(2)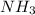 = 17.031g/mol
= 17.031g/mol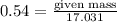
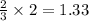 moles of ammonia.
moles of ammonia.