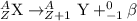Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 23.06.2019 01:00
The primary products of complete combustion of fossil fuels are a. carbon dioxide and water b. methane and water c. carbon monoxide and water d. carbon dioxide and carbon monoxide
Answers: 1


Chemistry, 23.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 4.20 mol fe and 6.70 mol nio(oh) react?
Answers: 3
You know the right answer?
What is the mass number of the particle emitted from the nucleus during beta minus (β–) decay?...
Questions


Computers and Technology, 08.03.2021 23:40

Mathematics, 08.03.2021 23:40

Biology, 08.03.2021 23:40


Mathematics, 08.03.2021 23:40



Computers and Technology, 08.03.2021 23:40

Mathematics, 08.03.2021 23:40

Arts, 08.03.2021 23:40

Mathematics, 08.03.2021 23:40

Mathematics, 08.03.2021 23:40



Social Studies, 08.03.2021 23:40

History, 08.03.2021 23:40


Chemistry, 08.03.2021 23:40