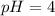Chemistry, 29.07.2019 03:00 itzyoboyCj
Asolution with a ph = 13 has approximately how many moles of oh– ions per liter? how many molecules of h+ would this same solution have per liter? a different solution with an h+ concentration of 1.0 × 10–4 would have a ph =

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which of the following statements is true? a. elements in the last period are radioactive. b. atomic weight is the same as atomic mass. c. elements in the same group have the same number of electron shells. d. atomic number equals the number of neutrons in the nucleus of an atom.
Answers: 1

Chemistry, 22.06.2019 01:00
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1

Chemistry, 22.06.2019 04:30
Acamcorder has a power rating of 17 watts. if the output voltage from its battery is 7 volts, what current does it use?units:
Answers: 1

Chemistry, 22.06.2019 07:00
How many moles are in 7.2 x 10^23 carbon molecules? (*round to the nearest hundredth and include the unit "mol c" after your number) question 6 options:
Answers: 2
You know the right answer?
Asolution with a ph = 13 has approximately how many moles of oh– ions per liter? how many molecules...
Questions

Mathematics, 11.02.2021 03:10


Mathematics, 11.02.2021 03:10

Mathematics, 11.02.2021 03:10

Mathematics, 11.02.2021 03:10

English, 11.02.2021 03:10

Mathematics, 11.02.2021 03:10


Geography, 11.02.2021 03:10



Biology, 11.02.2021 03:10

Mathematics, 11.02.2021 03:10

Mathematics, 11.02.2021 03:10

Mathematics, 11.02.2021 03:10


History, 11.02.2021 03:10

Health, 11.02.2021 03:10

Mathematics, 11.02.2021 03:10

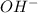 ions per liter is, 0.1 moles/L
ions per liter is, 0.1 moles/L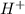 ion is,
ion is, 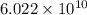
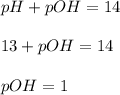
![pOH=-\log [OH^-]\\\\1=-\log [OH^-]](/tpl/images/0145/1283/6dfe1.png)
![[OH^-]=0.1moles/L](/tpl/images/0145/1283/cb909.png)
![pH=-\log [H^+]\\\\13=-\log [H^+]](/tpl/images/0145/1283/4a04f.png)
![[H^+]=10^{-13}moles/L](/tpl/images/0145/1283/cb161.png)
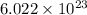 number of molecules of
number of molecules of 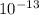 moles contains
moles contains 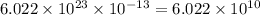 number of molecules of
number of molecules of ![pH=-\log [H^+]\\\\pH=-\log (1\times 10^{-4})](/tpl/images/0145/1283/9c3d6.png)