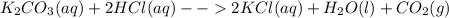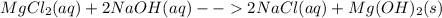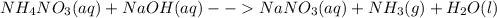Chemistry, 28.07.2019 04:40 ljdavies51
Predict what would be observed (and why) from an aqueous mixture for each of the following solution (all substances are water soluble).

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 10:00
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2


Chemistry, 22.06.2019 19:30
Acetylene gas c2h2 undergoes combustion to produce carbon dioxide and water vapor how many grams of water are produced by the same amount of c2h2?
Answers: 2
You know the right answer?
Predict what would be observed (and why) from an aqueous mixture for each of the following solution...
Questions

History, 14.12.2020 22:40


Biology, 14.12.2020 22:40


Social Studies, 14.12.2020 22:40



History, 14.12.2020 22:40



Chemistry, 14.12.2020 22:40

Mathematics, 14.12.2020 22:40


Health, 14.12.2020 22:40





Mathematics, 14.12.2020 22:40

English, 14.12.2020 22:40