
Chemistry, 27.07.2019 17:40 Shamplo8817
Which statement correctly describes the ka of a weak acid? the ka is large, and the equilibrium favors products. the ka is large, and the equilibrium favors reactants. the ka is small, and the equilibrium favors products. the ka is small, and the equilibrium favors reactants.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
The balanced chemical equation for this lab is: 3cucl2(aq) + 2al(s) 3cu(s) + 2alcl3(aq) if 10.5 g copper chloride react with 12.4 g aluminum, what is the limiting reactant?
Answers: 3

Chemistry, 21.06.2019 23:30
Start an single atom tab. observe the decay of polonium-211. after each decay, press the reset nucleus button to watch the process again. write a description of alpha decay for po-211
Answers: 2

Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

You know the right answer?
Which statement correctly describes the ka of a weak acid? the ka is large, and the equilibrium fav...
Questions


Physics, 14.09.2019 07:30






Physics, 14.09.2019 07:30





Mathematics, 14.09.2019 07:30




Mathematics, 14.09.2019 07:30

Mathematics, 14.09.2019 07:30


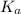 is small, and the equilibrium favors reactants.
is small, and the equilibrium favors reactants.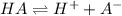
![K_a=\frac{[H^+][A^-]}{[HA]}](/tpl/images/0757/8147/66f51.png)
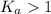 ; the reaction is product favored.When
; the reaction is product favored.When 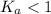 ; the reaction is reactant favored.When
; the reaction is reactant favored.When 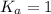 ; the reaction is in equilibrium.
; the reaction is in equilibrium.

