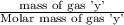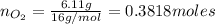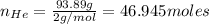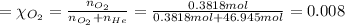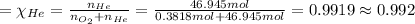Answers: 2
Another question on Chemistry



Chemistry, 22.06.2019 07:20
Part b: study of equilibrium on solubility: mg(oh)2(s) ⇌ mg2+(aq) + 2 oh–(aq) cloudy clear (pink) 7. a. b. 8. a. b. 9. 10. 11. 12. when adding concentrated hydrochloric acid, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 7a. you should indicate which ion was added to or removed from the equilibrium mixture. when adding edta, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 8a. you should indicate which ion was added to or removed from the equilibrium mixture. upon heating in which direction is the equilibrium shifting? upon cooling in which direction is the equilibrium shifting? is the forward reaction a. endothermic explain your answers to questions 9, 10, and 11. (l) left (r) right (l) left (r) right b. exothermic
Answers: 1

Chemistry, 22.06.2019 13:50
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2
You know the right answer?
Agas mixture called heliox, 6.11% o2 and 93.89% he by mass, is used in scuba tanks for descents more...
Questions




Mathematics, 15.12.2019 18:31


Mathematics, 15.12.2019 18:31

Mathematics, 15.12.2019 18:31

History, 15.12.2019 18:31


Mathematics, 15.12.2019 18:31

Mathematics, 15.12.2019 18:31


English, 15.12.2019 18:31


Mathematics, 15.12.2019 18:31


Business, 15.12.2019 18:31

Mathematics, 15.12.2019 18:31


English, 15.12.2019 18:31

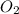 in the mixture is 0.008.
in the mixture is 0.008.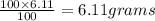
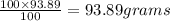
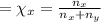
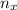 = moles of gas 'x'=
= moles of gas 'x'=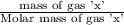
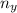 = moles of gas 'y'=
= moles of gas 'y'=