
Chemistry, 22.07.2019 10:30 dontworry48
What is kclo4 a weak acid, weak base, acidic salt, basic salt, or a neutral salt

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1

Chemistry, 22.06.2019 22:00
4.25g sample of solid ammonium nitrate dissolves in 60.0g of water in a coffee-cup calorimeter, the temperature drops from 22.0 c to 16.9 c. assume that the specific heat of the solution is the same as that of pure water. calculate delta(h) (in kj/mol nh4no3) for the solution proces.
Answers: 2

Chemistry, 23.06.2019 00:50
What is the enthalpy of combustion (per mole) of c4h10 (g)? –2,657.5 kj/mol –5315.0 kj/mol –509.7 kj/mol –254.8 kj/mol
Answers: 1
You know the right answer?
What is kclo4 a weak acid, weak base, acidic salt, basic salt, or a neutral salt...
Questions







Spanish, 11.01.2021 20:00

Mathematics, 11.01.2021 20:00

Biology, 11.01.2021 20:00

Arts, 11.01.2021 20:00


Mathematics, 11.01.2021 20:00

Mathematics, 11.01.2021 20:00


Mathematics, 11.01.2021 20:00

Spanish, 11.01.2021 20:00


Biology, 11.01.2021 20:00

Arts, 11.01.2021 20:00

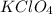 is formed when KOH ( Potassium hydroxide ) and
is formed when KOH ( Potassium hydroxide ) and 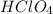 ( generally know as Perchloric Acid ) which is strong acid react with each other.
( generally know as Perchloric Acid ) which is strong acid react with each other.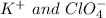 in which
in which 


